Dive Support Vessel
Commercial diving involves a diverse group of individuals and companies involved in a wide range of activities. These workers are exposed to the same hazards anyone would if they spent extended periods of time underwater, such as drowning, respiratory and circulatory problems, and hypothermia. The number of dives, length of time spent underwater, lack of visibility, and the strenuous nature of the task increase the risk of this type of activity. Additionally, commercial divers are often exposed to construction or demolition type hazards such as cutting, welding, material handling, cleaning, operation of heavy equipment, and general work with power tools.
SCUBA literally stands for Self-Contained Underwater Breathing Apparatus meaning that the diver carries all the needed breathing equipment and gases with them, and is subject to the water temperature, pressure, currents, and other environmental factors present at their diving depth.
The primary difference between open circuit and closed circuit breathing apparatuses is what happens to the exhaled gas. In open circuit diving, the diver's breathing gas is exhaled directly into the water; where as, in closed circuit diving, the diver's breathing apparatus recycles the diver's exhaled breath by removing the carbon dioxide and adding oxygen to replace the consumed oxygen. By recycling the diver's breathing gas, closed circuit breathing apparatuses allow the diver to be more streamlined and reduce the amount of gas tanks required.
Limiting factors such as, diving depth, gas mixtures and supply, weather, and decompression obligations can have a significant impact on the amount of time a scientist will actually have to conduct their research underwater. Saturation diving, a technique developed by the U.S. Navy in the 1950s, has proven to be useful to extend underwater work time. Saturation diving works on the premise that if a diver's tissues are in equilibrium with the surrounding water, then the decompression time will not change for the length of time spent underwater. This "saturation" process takes approximately 24 hours and means that the diver needs to remain at the same depth.
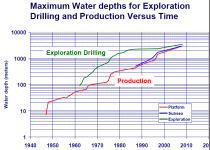
Atmospheric Diving Systems (ADS) are used by commercial and military customers who require a manned presence at the underwater work site. These one-man suits keep the operator at one atmosphere pressure at all times, and are extremely flexible and dexterous to permit work and structural inspections in tight work spaces. The HARDSUITST product line has constantly evolved since 1986, and the HARDSUITT1200 and HARDSUITT 2000 systems represent the state of the art in atmospheric diving. These systems provide a unique capability to offer deep diving capability without the high cost associated with saturation diving. The ability to have man's senses, perception and diver like agility at depths up to 2,000 feet without the physical risks of decompression provides an unparalleled resource to accomplish difficult subsea tasks in a safe, efficient, and low cost manner.
In August 2006 the Navy Hardsuit 2000, or Atmospheric Diving System, had its first test run, and it brought a Sailor to 2,000 feet below the sea without imploding. The suit took 11-years to develop, and is meant to scout out vessel damage on submarine rescue missions. Older suits only dove to 1,200 feet.
ROVs are unmanned underwater robots that are controlled by a pilot, via a long tether that is spooled out from the support ship. These robots can be fitted with advanced camera, lighting, and sampling systems allowing scientists to be virtually transported, through real-time video transmission, to great depths of the oceans. The advantages of ROVs include greatly extended bottom times, reduced human risk, more affordable technology, and the ability to deploy in harsher environments.
Of the 116 occupational diving fatalities reported by OSHA for 1989-1997 (13 deaths per year), 49 (five per year) occurred among an estimated 3000 full-time commercial divers. The average of five deaths per year corresponds to a rate of 180 deaths per 100,000 employed divers per year, which is 40 times the national average death rate for all workers. This group, which accounts for most of the commercial dive time underwater, includes divers involved in construction, maintenance, and inspection of vessels and structures such as oil rigs, bridges, and dams. The remaining 67 deaths occurred among workers who were not full-time divers; these include seafood harvest divers, search and rescue divers, scientific divers, dive instructors, and nonmilitary federal agency divers. NIOSH's National Traumatic Occupational Fatalities database reported 56 occupational diving deaths for 1989-1994 (11 deaths per year); causes of deaths listed most often for divers included drowning (73% of cases), asphyxia (14%), and embolism (7%). Other causes included trauma, hypothermia, and late medical complications, but hypothermia and air embolus may be underestimated because of difficulties in diagnosing these conditions.
Commercial divers are exposed not only to the possibility of drowning but also to a variety of occupational safety and health hazards such as respiratory and circulatory risks, hypothermia, low visibility, and physical injury from the operation of heavy equipment under water. The type, length, frequency of dive and the type of operation increase the already high risk of this strenuous work. Additional hazards are also associated with the actual work, such as underwater cutting and welding, materials handling, hull scrubbing, and use of hand and power tools.
A common type of health hazard associated with commercial diving is "Dysbarism". Dysbarism is a generic term applicable to any adverse health effect due to a difference between ambient pressure and the total gas pressure in tissues, fluids, or cavities of the body. Divers face an array of unique health hazards. The first two effects below are associated with hyperbaric conditions; the latter three occur due to changing pressures:
Other hazards presented to divers include microbes and parasites, noise, fire and chemicals during underwater cleanup operations.
Most dysbarisms are predictable using some combination of Boyle's Law, Dalton's Law, and/or Henry's Law. Boyle's Law applies to the expansion and contraction of gases within the body because of external pressure changes due to depth. Dalton's Law states that in a mixture of n gases the partial pressure of each gas [Pi] is proportional to the molar fraction of each gas [Yi] that makes up the total, and can be used to determine how much nitrogen, oxygen, or carbon dioxide is in the ambient air at any hyperbaric pressure underwater. Henry's Law states a gas will dissolve into a liquid in proportion to its partial pressure in the air and its solubility in the liquid, and can predict the body's absorption of inert gases into and back from the body at any pressure or depth.
Pain and barotrauma from expanding or contracting gases while transiting between pressure zones are the most direct effects predictable from Boyle's Law. The most common sites of pain from trapped gases are the digestive tract, sinuses, teeth, middle ear, and lungs (the latter particularly during ascent).(1,3,7,16) Good dental care should avoid trapped gas in teeth. Divers and flyers should anticipate these effects and not attempt to suppress the release of natural digestive gases that will expand during ascent. Blockage of sinus passages due to nasal congestion or a head cold can cause pain during either ascent or descent. Sinus pain during descent is called "sinus squeeze." Divers should be trained to detect blocked sinuses and not dive with a cold or an allergic inflammation. The most common source of pain on descent is from the contraction of air in the middle ear if the eustachian tubes are inflamed or blocked. Divers should be trained to clear their ears every two feet, and to stop and rise back up a few feet before attempting to clear a blockage.(1,13) The most severe outcome of expanding gases is pulmonary barotrauma. An increase in gas volume of 20 to 30 percent can cause an initially full lung to rupture. It is important to realize that the proportionate change in pressure and trapped gas volume (predictable by Boyle's Law) occurs over smaller distances at shallow depths than when starting from deeper depths.
Decompression sickness (DCS) is the most commonly known of the many dysbarisms. It is sometimes referred to as "evolved gas dysbarism," "compressed air sickness," "caisson worker's syndrome," or various common names listed in Table 2. DCS is completely different from the preceding direct effects. DCS is caused indirectly by the formation of inert gas bubbles (nitrogen or helium if used) at one or more locations within the body, corresponding to the symptoms. The gas is absorbed slowly at depth from the blood to the tissues. The amount of gas absorbed depends upon depth and "bottom time."(1,3) During ascent, divers can easily decompress to lower pressures at rates much faster than the stored gases can be reabsorbed back into the blood and exhaled out of the body. If the pressure ratio is too large, bubbles form and symptoms of DCS occur, ranging from mildly irritating to severe. A simple medical classification of DCS has evolved. Type I DCS consists of only skin, lymphatic, or joint pain. DCS Type II involves respiratory symptoms, neurologic or auditory-vestibular symptoms, and shock or barotrauma symptoms. Type II DCS is potentially life-threatening. The incidence of DCS is largely unknown.
Scientific Diving is done by those individuals needing to perform dives to collect scientific data. Examples include: collecting (water, bottom samples, flora and fauna, etc.), observing and documenting (including the use of cameras), and measuring and counting. Another way to describe this classification is "limited." The tasks of a scientific diver are limited to observation and data gathering and are performed in advancement of science. If tools are used, they are limited to "light" hand tools (e.g., small hammers, pliers, screw drivers, chisels, pneumatic-power drills, etc.). Other ancillary equipment such as small lift bags (< 50 pounds lifting capacity) and small air lifts can also be used if associated with data collection. Tasks typically associated with commercial diving, such as lifting heavy objects, construction, use of heavy hand or power tools, ship husbandry tasks are not authorized under the scientific diver classification. Tasks performed by scientific divers are exempt from the OSHA commercial diving regulations.
In comparison to the scientific classification, the working diver is "unlimited" by the tasks to be performed. Examples of working tasks include: ship husbandry (cleaning hulls, sea strainers, replacing zincs, unfouling a ship's propeller), use of heavy power tools powered by pneumatics or hydraulics from the surface, lifting and positioning heavy objects weighing >50 pounds, locating and retrieving items, construction, and underwater cutting using exothermic cutting lances. Although working divers can perform both scientific and/or commercial diving tasks, when performing the latter, they are not exempt from the OSHA commercial diving regulations.
In 1984 the distributions of fatal diving accidents in commercial diver populations were examined in the Gulf of Mexico from 1968 to 1975 and in the British sector of the North Sea from 1971 to 1978. Influences and causes of death were analyzed by examining the interaction between host, environmental and agent factors. The interaction of host and environmental factors appeared to be the greatest contributing factor to diving fatalities among the estimated 900 commercial divers in the Gulf of Mexico and the 700 in the North Sea. The most significant host factors were level of experience and behavioral dysfunction. They are also the host characteristics most amenable to change through improved and more thorough training. The most significant environmental factors were equipment failure and supervisor/tender errors. These factors would be minimized by improved selection, maintenance and operation of equipment, together with improved operating and emergency diving procedures. In recent years there has been a significant downward trend in mortality rates in the commercial diver populations of this study due to improved diving techniques and operations. Further research is needed, however, on the cause(s) of diver unconsciousness and inexplicable actions that occur at depths below 90m (300 ft.).
|
NEWSLETTER
|
| Join the GlobalSecurity.org mailing list |
|
|
|